At pressures above about 20 atm, 2 effects start playing a role. First, the active pressure, or fugacity, of the gas becomes different from its (partial) pressure:
[PCO2] = φCO2 PCO2 / P0,
where φ is the fugacity coefficient. The gas pressure is divided by the standard state (P0 = 1 atm) to make the [activity], or [fugacity] dimensionless.
Second, the pressure affects Henry's constant by the volume change of the reaction (as explained in Pressure effects on solubility).
The fugacity coefficient can be obtained by comparing an equation of state for the gas with the ideal gas law. For example, the Van der Waals equation:
where b is the gas' minimal volume, and a and α are Van der Waals attraction factors.
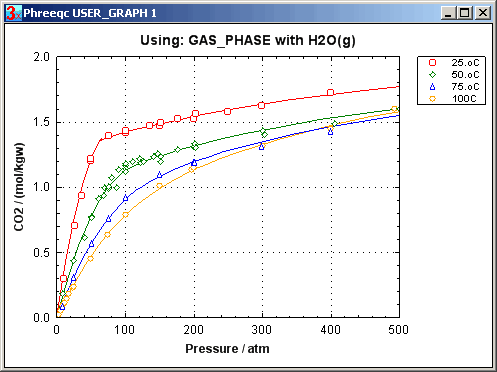
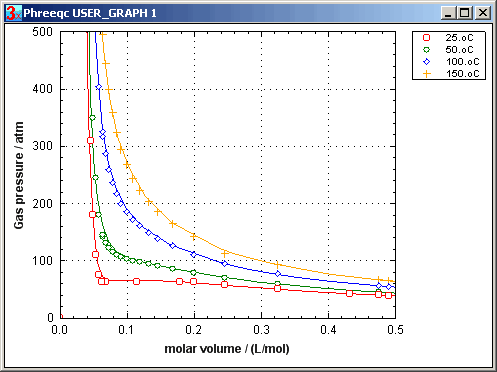
Peng and Robinson (1976) have devised formulas to relate these coefficients to the critical pressure and temperature, and the acentric factor of a gas. The Peng-Robinson formulas enable a remarkably accurate estimate of the P-Vm relationship for a gas. Calculated (lines) and measured (symbols) data on CO2 by Michels et al. (1935, 1937) are compared in the graph (PHREEQC input file CO2_VP.phr). Note how the P-Vm curve at 25oC flattens out, when the gas is compacted to 0.17 L/mol, where the gas becomes liquid. When, at 0.065 L/mol, all the gas has been liquefied, further compaction results again in pressure increase. Above the critical temperature of 31oC, liquid and gas are indiscernible, and the constant-pressure effect is missing.
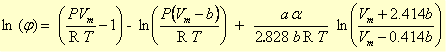
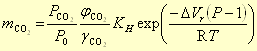 .
.